Nationwide Trends
All Payer Claims Databases (APCD)
For more on APCD’s, check out the report “The Secret of Health Care Prices: Why Transparency Is in the Public Interest”.
Surprise/Balance Billing Protections
For more on state surprise billing statutes, refer to our Issue Brief on The Source Blog.
State Merger Review Authority
For more on provider and insurer market consolidation, see key issue page Market Consolidation.
State Regulation of Anticompetitive Provider Contract Terms
See major litigation and resource tables on key issue page Provider Contracts.
Certificate of Public Advantage Laws
For more on provider and insurer market consolidation, see key issue page Market Consolidation.
Telehealth Requirements Under Existing Mandates and COVID-19 Emergency
See data charts on COVID-19 Crisis Key Issue Page for more information.
State Public Option Proposals
For more on state public option legislation, see key issue page System Reform.
The State Database
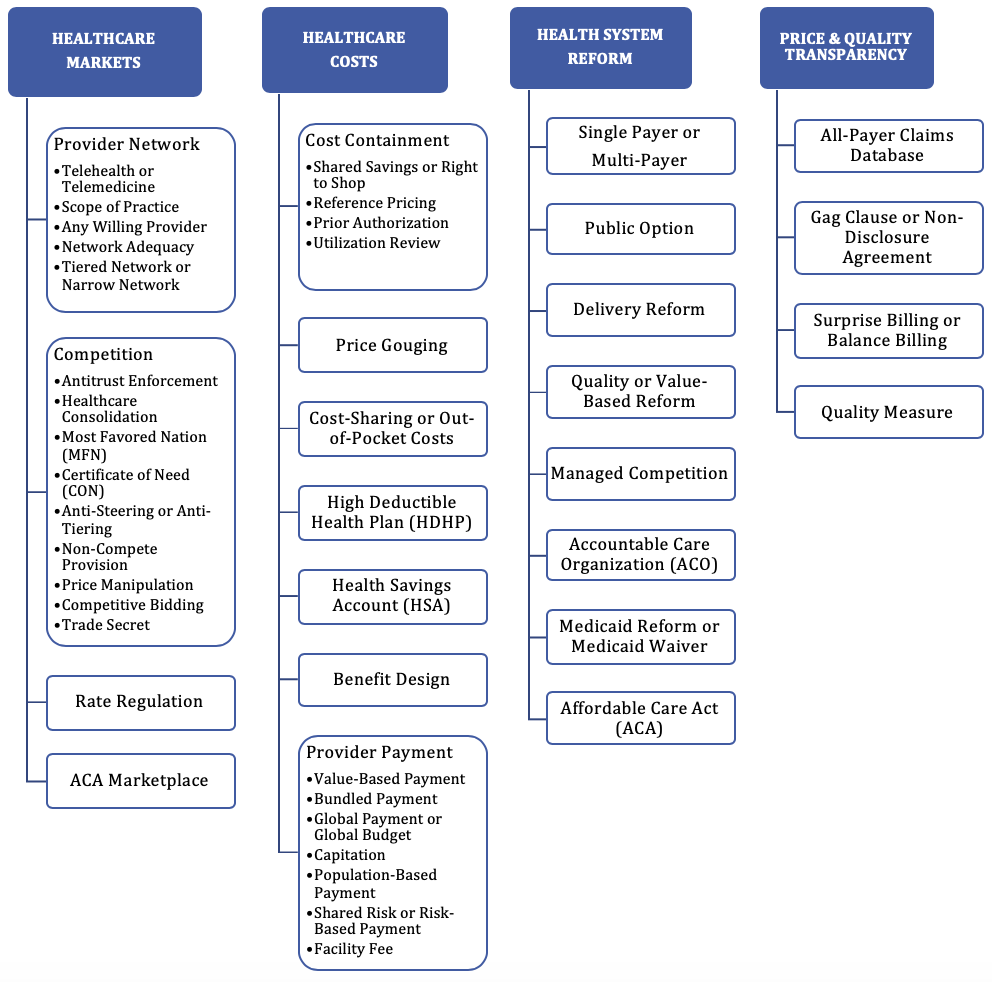
State laws and regulations governing healthcare can vary greatly and have the potential to impede or facilitate improvements to the functioning of the healthcare marketplace. The Source tracks state activities impacting healthcare price and competition in both legislation and litigation in a searchable database to help stakeholders at the state level understand their legal and regulatory environment as they make efforts to improve access, quality, and efficiency, and reduce costs in healthcare.
LEGISLATION: The Database of State Laws Impacting Healthcare Cost and Quality (SLIHCQ), created by The Source on Healthcare Price & Competition and Catalyst for Payment Reform with support from the Robert Wood Johnson Foundation, catalogues state legislation to contain healthcare costs and improve quality in a searchable and sortable, public database. These laws impact price transparency, provider market power, provider payment, provider networks, and benefit design. The database also includes pharmaceutical legislation beginning in the 2017-2018 legislative session, and continues to populate newly enacted pharmaceutical legislation as part of the database.
*Note: Current legislative session bill updates are ongoing. Check back weekly for updates.
LITIGATION: The Source tracks major litigation and antitrust enforcement action by federal entities (FTC or DOJ), state attorney generals, and private parties in the main provider and insurer markets, particularly legal challenges of healthcare consolidation and anticompetitive contract provisions. Additionally, the database contains major pharmaceutical cases including legislation challenges and significant appellate cases.
USER GUIDE: The Database utilizes key issue tags for all legislation and litigation records as a guide for users to quickly identify important topics (see User Guide for more info). Search the Database for specific bills, statutes or cases by key issue category, keyword, and/or jurisdiction. *Multiple filter/selections enabled.
111 results returned.
District Court: District of New Jersey District Court Docket: Status: Decided
Consumers brought putative class action against manufacturers and distributors of prescription eye medication, alleging they packaged medication in such a way as to force consumers …
Drogueria Betances LLC v. Novartis Pharmaceuticals Corp.
District Court: Southern District of New York District Court Docket: Status: Pending
Novartis Pharmaceuticals Corp. allegedly agreed with Par Pharmaceuticals Inc. to squelch generic competition to Novartis’s hypertension drug Exforge, resulting in buyers paying millions more than …
Federal Trade Commission v. AbbVie Inc. et al.
District Court: Eastern District of Pennsylvania District Court Docket: Status: Decided
FTC alleged AbbVie sued generic companies for patent infringement over their proposed generic version of the testosterone gel, AndroGel, without justifiable grounds but merely to …
In the Matter of Impax Laboratories Inc.
District Court: Federal Trade Commission District Court Docket: Status: Pending
The Federal Trade Commission sued generic drugmaker Impax Laboratories Inc. for allegedly accepting more than $100 million to delay its release of a generic version …
In Re: Generic Pharmaceuticals Pricing Antitrust Litigation
District Court: Eastern District of Pennsylvania District Court Docket: Status: Pending
Plaintiffs are attorney generals from 48 states, Puerto Rico, and the District of Columbia, as well as classes of private plaintiffs that filed an antitrust …
Previous | Next | See all Litigation Records
Individual State Pages
If you would like to report a possible data discrepancy, please email info@sourceonhealthcare.org or contact The Source here.

